Pharmacological Characterization of the RPMI 2650 Model as a Relevant Tool for Assessing the Permeability of Intranasal Drugs | Molecular Pharmaceutics

Unusual Growth and Hydration Characteristics of Oil Solubilized Micelles in Aqueous Pluronic Systems | The Journal of Physical Chemistry B
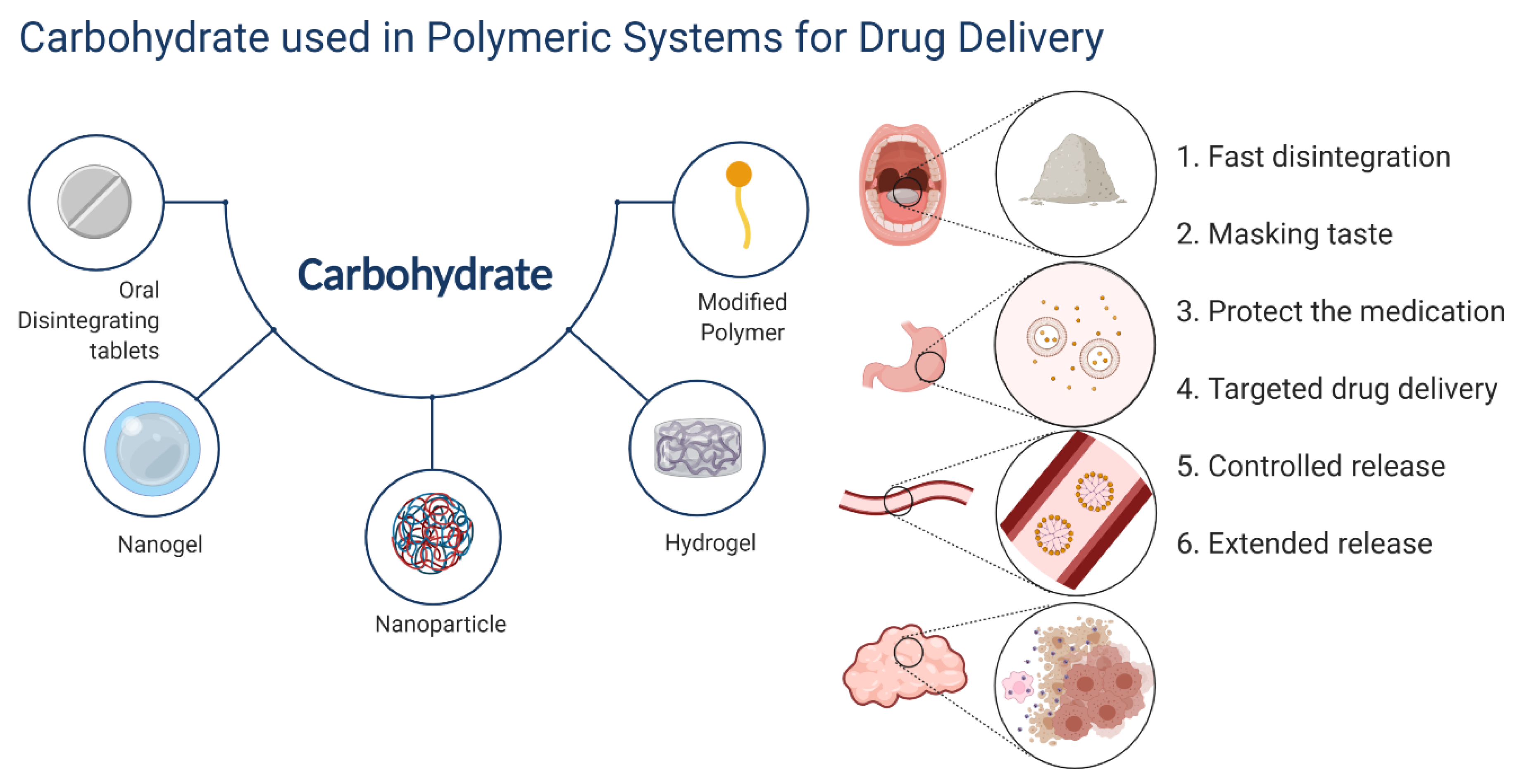
Pharmaceutics | Free Full-Text | Carbohydrates Used in Polymeric Systems for Drug Delivery: From Structures to Applications

Medica RCP |Unimer Pediatric Hypertonic | Indications | Side Effects | Composition | Route | all.price | Alternative Products